 For the molecule H3O+ determine the following: Molecular geometry -Select- linear, trigonal planar, bent, tetrahedral, trigonal pyramidal, trigonal bipyramidal, T-shaped, seesaw. In ammonia the trigonal pyramid undergoes rapid nitrogen inversion. The F-S-F axial plane forms a 173-degree angle with an length of 3.28 Angstroms, which can be attributed to the lone pair of electrons on the S atom. Question: In this question you will be asked to draw several Lewis structures and determine the geometry, bond angle, and hybridization about the central atom. 
In contrast, boron trifluoride is flat, adopting a trigonal planar geometry because the boron does not have a lone pair of electrons. However, the three hydrogen atoms are repelled by the electron lone pair in a way that the geometry is distorted to a trigonal pyramid (regular 3-sided pyramid) with bond angles of 107°. This would result in the geometry of a regular tetrahedron with each bond angle equal to cos −1(− 1 / 3) ≈ 109.5°. The nitrogen in ammonia has 5 valence electrons and bonds with three hydrogen atoms to complete the octet. 
Phosphine, an example of a molecule with a trigonal pyramidal geometry. Use the VSEPR shape to determine the angles between the electron domains. Use the steric number and VSEPR theory to determine the electron domain geometry of the molecule. Write the Lewis dot structure for the molecule. 9: (a) H 2 O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. The bond angle can easily find by using the VSEPR theory. The AXE method for VSEPR theory states that the classification is AX 3E 1. Thus, the electron-pair geometry is tetrahedral and the molecular structure is bent with an angle slightly less than 109.5°. c) CH3I is tetrahedral, but not symmetrical, making us uncertain of. In organic chemistry, molecules which have a trigonal pyramidal geometry are sometimes described as sp 3 hybridized. b) BCl3 is trigonal planar and symmetrical, making us confident the bond angles are 120. Some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (XH 3), xenon trioxide (XeO 3), the chlorate ion, ClO −ģ. The molecular geometry, or three-dimensional shape of a molecule or polyatomic ion, can be determined using valence-shell electron-pair repulsion (abbreviated VSEPR and pronounced VES-per) theory, in which the basic principle is valence electrons around a central atom stay as far apart as possible to minimize the repulsions. When all three atoms at the corners are identical, the molecule belongs to point group C 3v. 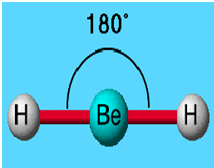
In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry). The bond angle indicated is exactly 109. Which of the following is the best statement about the bond angle indicated by the double arrow in the diagram. Molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base Trigonal pyramidal molecular geometry Transcribed Image Text: Part A Below is a cartoon drawing of a seesaw molecular geometry which is based upon the trigonal bipyramidal electron group geometry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
